A phase 2 trial has shown that a 2-week course of triple antiviral therapy with (1) interferon beta-1b (used to treat the relapsing-remitting and progressive forms of multiple sclerosis), (2) lopinavir-ritonavir (for the treatment of HIV/AIDS) and (3) ribavirin (used to treat chronic hepatitis C and other flavivirus infections) is safe and better at shortening COVID-19 viral shedding than lopinavir-ritonavir alone (average 7 days vs. 12 days) in patients with mild to moderate illness if treatment is started within 7 days of symptom onset.
The study is registered with ClinicalTrials.gov, NCT04276688.
A Phase 3 study needs to be done and submitted to regulatory agencies for this regimen to be approved. Phase 3 study is a large, properly powered human clinical trial, typically featuring hundreds or thousands of people. To move forward with the trial, investigators need to demonstrate that the medication is at least as safe and effective as existing treatment options. The phase 3 trial will be designed to evaluate whether this specific 3-drug combo is better than the other treatment, 2-drug combo, in patients infected with SAR-CoV-2, the coronavirus causing COVID-19.
The prospective study, published in The Lancet, involved 127 adult COVID-19 patients (mean age, 52 years) admitted to six Hong Kong hospitals from February 10 to March 20, 2020. Eighty-six patients were randomly assigned to receive 2 weeks of the triple-drug regimen every 12 hours plus as many as three doses of injectable interferon beta-1b every other day, while 41 received lopinavir-ritonavir alone every 12 hours.
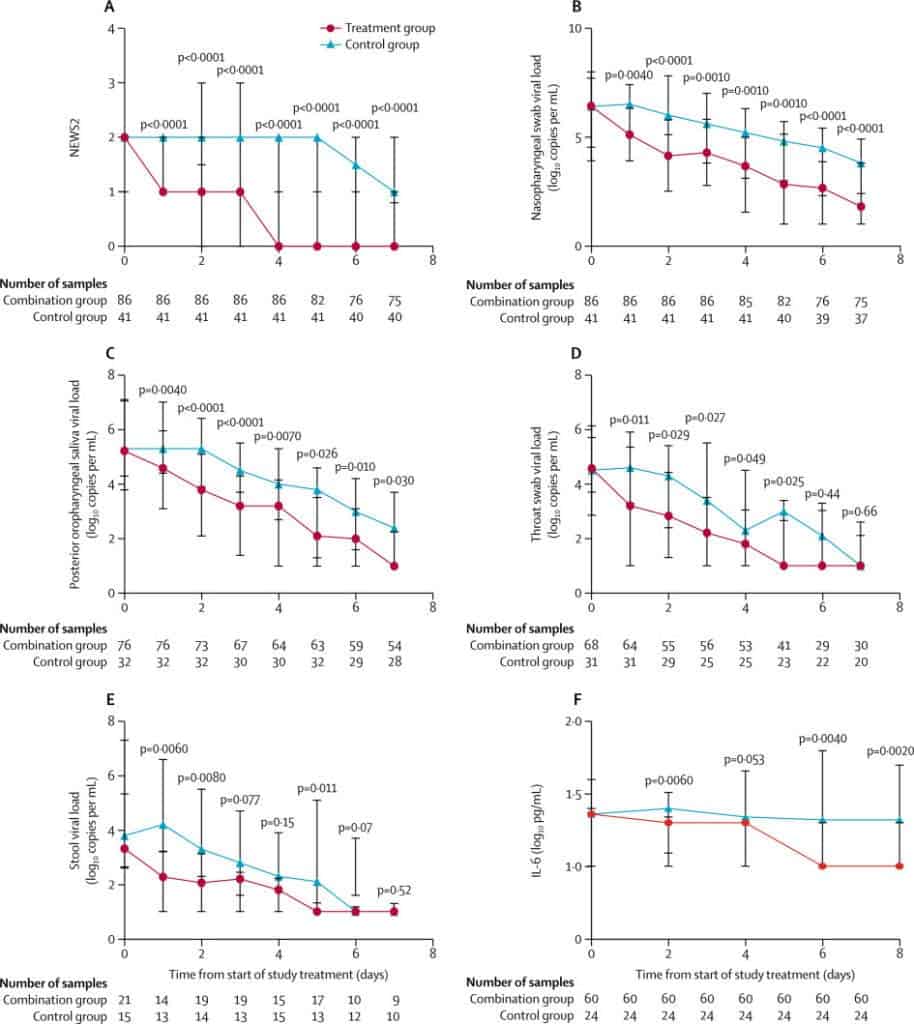
The investigators detected no live coronavirus on nose-throat swabs within, on average, 7 days after the triple-drug combination started, 5 days earlier than with lopinavir-ritonavir alone (hazard ratio, 4.37; 95% confidence interval, 1.86 to 10.24; P = 0.0010). The 3-drug combination also reduced the mean time to complete symptom relief into half (4 versus 8 days) and Sequential Organ Failure Assessment score of 0 (3 versus 8 days) and shortened the mean hospital stay from 14.5 to 9 days.
The findings also suggest that interferon beta 1-b may be a key component of the combination and is worth further investigation for the treatment of COVID-19. Interferons are naturally occurring proteins, produced in response to viral infection and interferon beta-1b could potentially boost the body’s ability to fight SARS-CoV-2.
However, while patients who received the combination therapy within 7 days of symptom onset did better than those in the control group, there was no difference when they were treated 7 or more days after the start of symptoms. The researchers said that the results suggest that the 3-drug combination may minimize the risk of antiviral resistance and decrease risks to healthcare workers by reducing the duration and quantity of viral shedding.
Was this helpful?



