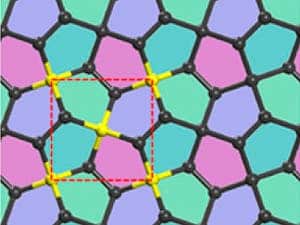
Chinese researchers ran simulations and found that a pentagon-containing version of graphene is theoretically stable. The 2D allotrope of carbon is made up of atom-thick sheet of carbon atoms arranged in a repeating pentagon pattern, while graphene is made up of carbon atoms arranged in a hexagon pattern, like a chicken wire. Graphene is the strongest material in the world and fantastic electrical conductor, among other wonder properties, but the penta-graphene would be even stronger and possibly more useful in electronics since it readily comes as a semiconductor. The “only” big challenge researchers face is actually synthesizing the 2D carbon pentagon pattern, and the Chinese team already has some ideas in mind. It’s worth considering that graphene was once thought to be impossible to synthesize.
Stronger, better graphene
Qian Wang, a researcher Peking University, first had the idea of studying penta-graphene after she saw an artwork on the wall of a Beijing restaurant depicting pentagon-shaped street tiles.
‘When I saw the wall decoration with a pentagonal tiling pattern, I conceived the idea of building a 2D carbon sheet using only pentagons,’ she says.
“I told my husband, “Come, see! This is a pattern composed only of pentagons,'” she said. “I took a picture and sent it to one of my students, and said, ‘I think we can make this. It might be stable. But you must check it carefully.’ He did, and it turned out that this structure is so beautiful yet also very simple.”
Inspired, Wang worked together with researchers from the University of Science and Technology of China in Hefei, Tohoku University in Japan and Virginia Commonwealth University in the US to see whether penta-graphene would be possible. Amazingly, not only did the simulations show that it is stable it should also be stronger than conventional graphene and be able to withstand higher temperatures, up to 730°C.
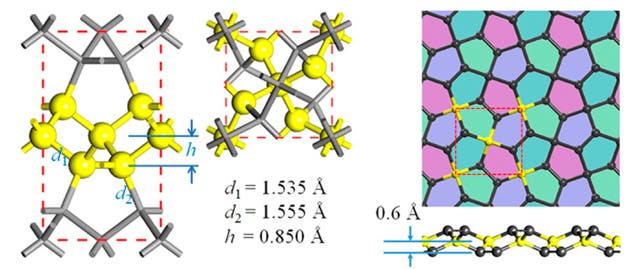
While it has very appealing mechanical properties, penta-graphene – if ever synthesized – might become a fantastic electronics feedstock. Graphene exhibits exceptionally high charge carrier mobility, which makes it appealing for future electronic applications. Unfortunately, it also has no band-gap, so we need to chemically alter it with impurities to turn it into a semiconductor. Penta-graphene on the other hand is a natural semiconductor. This could potentially prove useful for manufacturing carbon nanotubes (CNTs), as current CNT synthesis processes produce a mixture of metallic and semiconducting CNTs, depending on the orientation of the hexagons when a graphene sheet is rolled up. In contrast, rolling up a sheet of penta-graphene should only produce semiconducting CNTs. Penta-graphene is also auxetic, making it ideal for anything from electronic circuits to body armour.
‘If you stretch graphene, it will expand along the direction it is stretched, but contract along the perpendicular direction,’ explains Wang. ‘However, if you stretch penta-graphene, it will expand in both directions.’
In graphene, each carbon atom is sp2 bonded to 3 other carbon atoms, forming a 2-dimensional hexagonal lattice. In diamond, each carbon atom is sp3 bonded to 4 other carbon atoms, forming a 3-dimensional lattice. In this work, the authors describe a way of filling up a 2-dimensional plane with carbon atoms in which some are bonded to 3 neighbors and some are bonded to 4. The result is a buckled sheet (so not purely 2-D) of atoms that forms a tiling of non-regular pentagons (compared to graphene’s tiling of regular hexagons).
[RELATED] How to make graphene in a kitchen blender
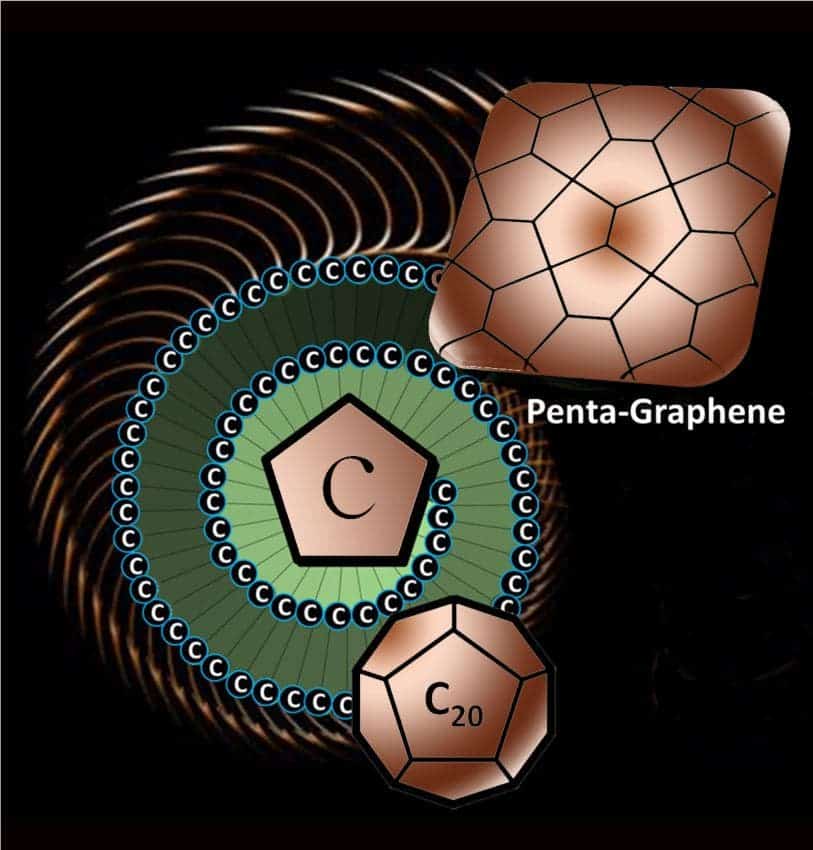
Some 3D materials contain both hexagon patterned and pentagon pattern carbon, like the spherical C60 buckyballs, but so far scientists have only been able to synthesise a single carbon nanomaterial made exclusively of pentagons in the form of a C20 cage. Wang and team suggest 2D penta-graphene could be harvested by peeling single sheets from a layered material known as T12-carbon – another purely theoretical carbon arrangement. The penta-hexagon simulation was reported in the journal PNAS.
‘We are looking for some experimental groups in China that are good at synthesising new materials,’ says Wang. ‘Penta-graphene is energetically more stable than C20 [and so] we hope that it will not be so difficult to synthesise.’