Although plasma is unstable in terrestrial conditions, it’s the most common state of matter in the universe, making up a large chunk of all the stars in the universe. It’s also pretty weird, posing many questions that researchers are still working to unravel.

We’re taught early in school that the basic states of matter are solid, liquid, and gas. There are other exotic states that scientists discovered more recently (like a superfluid or a Bose-Einstein condensate, for instance), but those three are what we learn as the “main” states of matter. However, your primary school teacher may have missed another one: plasma.
Plasma has a lot to do with heat: add enough heat to solids and they become liquid, next liquid becomes gas. Finally, with enough energy to ionize atoms into a soup of electrons and ions (electrically charged atoms), you have plasma.
Plasma has some unique characteristics that emerge as a result of the way particles interact with each other in this state. Let’s have a look at them.
Plasmon and Debye Shielding
Consider some electrically neutral plasma — this means that, between the charged free ions and electrons, positive and negative charges cancel each other out. If we change the position of a few electrons, even if it’s only a few, the displacement will change the electric equilibrium, so the now-unbalanced ions will try to move to a position to restore equilibrium. Because opposite charges attract, when the electrons move, the positive ions try to pull them back and make everything organized again. In plasma, this oscillation happens until some equilibrium is reached. This phenomenon is called Langmuir waves.
If you add charged particles to the plasma, the ions and electrons rearrange themselves in order to reach a nearly neutral state. To do so, electrons create a sort of electromagnetic shield around the positive particle, repelling the positive ions from it. This is called Debye Shielding.
The region necessary to involve such a particle is called a Debye sheath — or an electrostatic sheath. This type of sheath appears in plasma because the electrons tend to have a much higher temperature than that of the ions. This creates a layer in the plasma that has a greater density of positive ions, and hence an overall excess positive charge.
Depending on the charge and the characteristics of the plasma, the sheath can be bigger or smaller. Scientists classify plasmas based on the size of this sheath — an “ideal” plasma has lots of particles per Debye sheath volume, while fewer particles make it harder for electrons to shield new particles.
The early universe was made of plasma
Plasma can get very weird — which is why it’s somewhat surprising that the entire universe was, at some point, plasma.
During the first 10 to 15 microseconds of the universe, it was filled with a super hot soup made of particles called gluons and quarks. Gluons are the “glue” that sticks quarks together to form protons, neutrons, and other larger particles. During that early period, the universe was nearly 2 trillion Kelvin hot. This is the hottest the universe has ever been.

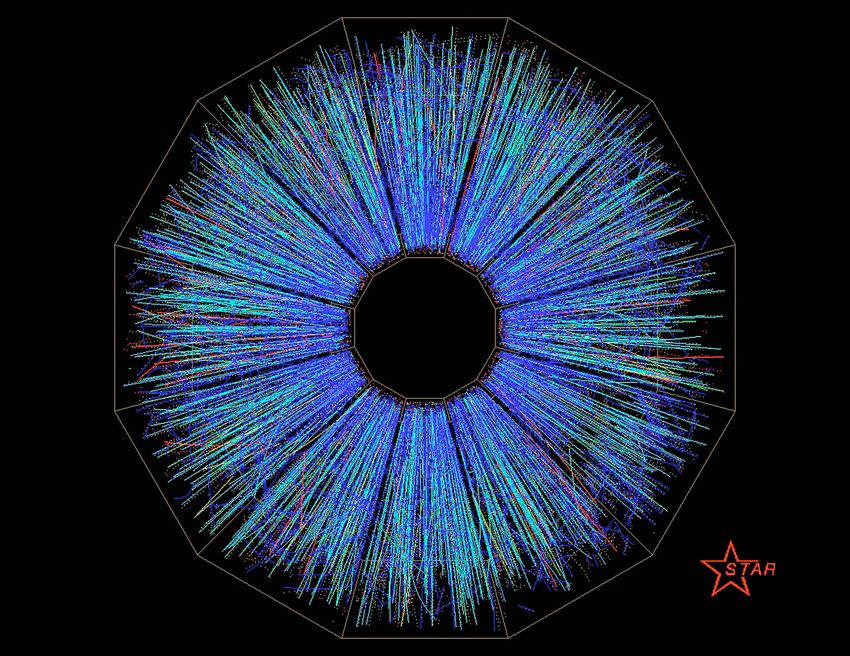
We can simulate such conditions in a particle collider. In these accelerators, scientists smash heavy gold or lead ions together to produce the quark-gluon soup (QGP) — the stuff that made up the early plasma universe. In such conditions, they learned that this state of matter behaves like a perfect fluid, not viscous like honey.
The two places perfect to form QGP are the Relativistic Heavy Ion Collider (RHIC) at Brookhaven National Laboratory and at the Large Hadron Collider (LHC) at CERN. The ions are accelerated to 99.995% of the speed of light, and QGP exists just for a very small fraction of a second (nearly zero) before it condenses again to form heavier particles.
Scientists collect the data from the accelerator and try to explain the mess with quantum chromodynamics- a very complex theory that strives to describe one of the fundamental interactions in nature — the strong interaction.
Starstuff
You don’t need to go into the lab to find plasma — simply go outside. We have the sun (during the day) and stars (during the night). On some days, you can even see it in the sky, in the form of lightning.
Plasma is also found scattered across the universe. Called the Warm–hot intergalactic medium (WHIM), there’s plasma of around a billion Kelvin found between galaxies. Around galaxies, there is a reservoir of diffuse gas in the form of plasma called the circumgalactic medium (CGM). This is usually hard for scientists to study because the gas has a very low density, but some simulations seem to try to understand the role of CGM in galaxy formation.
You may find plasma in planets as well, mostly in their magnetospheres – the regions of planets’ magnetic field affecting charged particles coming from space. The magnetospheres serve as protection from the solar wind, and in the case of the giant planets, these regions can be larger than the Sun. Inside Jupiter’s inside the dawn flank of the magnetopause, scientists found protons and heavy ions.

Plasma is a special state of matter, it is easily found in the universe. Unlike the other states of matter, it comes with other special properties because it is made of charged ingredients. Thanks to plasma physics, we understand the early stages of the universe and astrophysical objects.


