University of Pennsylvania and the University of California researchers have found a way to heal wounds and prevent scar tissue from forming. Newly identified growth factors, particularly Bone Morphogenetic Protein (BMP), instruct scar tissue-forming cells to transform into fat, regenerating the skin.
Healing without scars
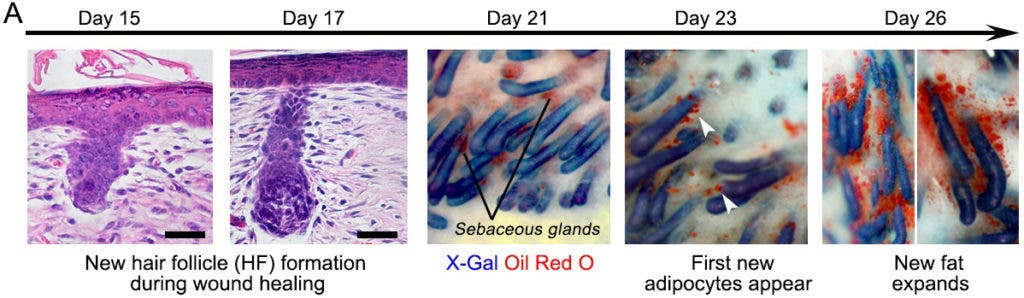
For many people, scar tissue is undesirable because of its unappealing looks. Scars themselves aren’t ugly — the discontinuity in an otherwise continuous smooth and familiar surface is. That’s because scar tissue lacks hair follicles and has a particular texture due to the absence of fat cells.
Myofibroblasts are one of the main cells found in healing wounds and their presence leads to scar formation. So, the researchers tried to find a way to turn these cells into something else, like adipocytes — the fat cells present in normal skin. Of course, that’s easier said than done. Previously, this sort of conversion was deemed impossible.
“Essentially, we can manipulate wound healing so that it leads to skin regeneration rather than scarring,” said George Cotsarelis, MD, the chair of the Department of Dermatology and the Milton Bixler Hartzell Professor of Dermatology at Penn, and the principal investigator of the project. “The secret is to regenerate hair follicles first. After that, the fat will regenerate in response to the signals from those follicles.”
The researchers first learned that hair and fat are produced separately but not independently. Knowing this, Cotsarelis and colleagues used a trial and error approach to discover the growth factors necessary for the formation of hair follicles. They then struck jackpot with the discovery of growth factors produced by the regenerating hair follicle that convert the surrounding myofibroblasts into fat. What happened was the skin became regenerated, instead of the wound turning into a scar tissue, as reported in the journal Science.
Because the fat is dependent on the new hairs, it will not form otherwise. However, the new cells are indistinguishable from the pre-existing fat cells, which makes the healed wound look like nothing happened instead of leaving a nasty, permanent impression on the skin.
“Typically, myofibroblasts were thought to be incapable of becoming a different type of cell,” Cotsarelis said. “But our work shows we have the ability to influence these cells, and that they can be efficiently and stably converted into adipocytes.” This was shown in both the mouse and in human keloid cells grown in culture.
“The findings show we have a window of opportunity after wounding to influence the tissue to regenerate rather than scar,” said the study’s lead author Maksim Plikus, PhD, an assistant professor of Developmental and Cell Biology at the University of California, Irvine.
The main application of such a therapy is obvious, as many people would love to have their wounds healed sans scaring. But there’s more potential to it than meets the eye. More fat cells in the tissue is desirable for some patients, like those suffering from HIV, who have adipocyte loss. Wrinkles, especially the very deep variety present on the skin, appear because adipocyte cells are lost to aging, so that’s another big demand waiting to be filled.
Researchers are now working on ways to make their findings transferable to the real world, a.k.a. turn it into a cream or something similar.
Was this helpful?