A new signaling mechanism that epithelial cells use to communicate during motion has been uncovered, explaining how individual (or groups of) cells can move inside of a tissue without compromising its structure.
One cell left to its own devices can move about pretty easily if needed. Its motion is similar to a ‘flow’ of sorts, with the cell’s leading edge extending a protrusion and its trailing edge drawing itself along. The same process of motion is used by cells included in tissues, with the extra requirement that the overall structure remains intact. So how do these tiny bits of life communicate and co-ordinate their motions in the context of tissues?
Protein nudges
When you need to pass through a tightly-packed group of people, your best bet is to communicate your intention of moving forward (verbally or through nudging) so they’ll make way. Cells in tissues do the same thing, but with proteins. Some of the signal proteins used in this process have been documented for some time now, but research from the University of Chicago has identified a new protein-driven signaling system that epithelial cells use to coordinate individual movements to move whole tissues at once.
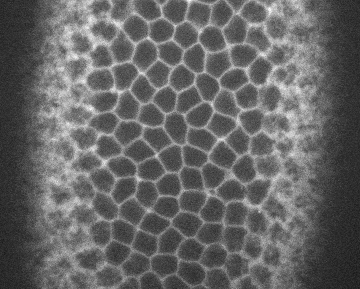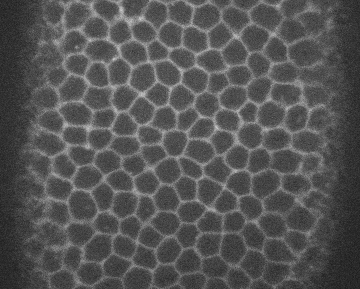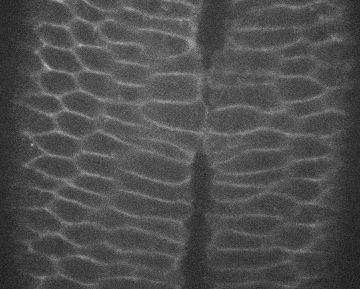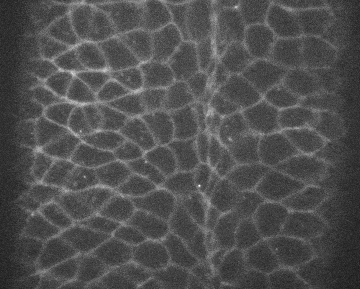
Image credits Institute Pasteur via giphy.
Cell biologist Sally Horne-Badovinac, PhD, and colleagues from the UoC found that two membrane proteins work in tandem to coordinate epithelial migration in Drosophila, the common fruit fly. The first one is called Lat, and works on the leading edge of the cell. The other, Fat2, acts at the trailing edge. Lets say we have three cells, A, B, and C, one behind the other, as the whole tissue needs to move.
For B to migrate, its Fat2 molecules first signal to the Lar behind it, which causes that cell (A) to extend its edge and go under B. In turn, B extends its leading edge under C, nudged by C’s Fat2. When A finishes its motion, its Lar signals to B’s Fat2, which retracts its leading edge — and the same happens between B and C.
Through this step-by-step process, neighboring cells can coordinate and move the whole tissue at the same time without leaving any holes in it.
“The protrusion of one cell goes underneath edge of the cell ahead, so you get what looks like overlapping shingles on a roof,” said Horne-Badovinac, an assistant professor of molecular genetics and cell biology and first author of the study.
“This process is understood really well at the single cell level, but when you hook these cells all together in a tight sheet, it becomes something more coordinated.”
The team used fruit flies to study this signaling process. As female embryos develop, the tissues which will later form egg chambers stretch and rotate into their final position. Scientists knew that both Fat2 and Lar were involved in this process, but it wasn’t clear if the cells were migrating because the tissues rotate around the circumference of a circular chamber, not moving in a straight line.
So Horne-Badovinac and her team grew the egg chambers in cell cultures outside of the female flies to get a better look at how they behaved. In groups of normal cells located behind cells edited to lack Fat2, the leading edge protrusions didn’t form. In groups of normal cells placed in front of Lar-missing patches, the trailing edges weren’t retracted.
“It was surprising, because what we knew was that the protein [Fat2] was at the trailing edge of the cell, but we were seeing an effect at the leading edge of the cell. So initially that made absolutely no sense,” said Horne-Badovinac.
“It required careful analysis along those cloned boundaries to really figure it out.”
Horne-Badovinac said there are still a lot of questions regarding the interaction between these proteins, and thinks there are other proteins which handle motion signaling to organelles inside the cell — especially the cytoskeletal machinery, which drives cellular movement.
Uncovering the mechanisms of coordinated cell movement could help us better understand critical stages of embryonic development, wound healing, and even cancer spread.
The full paper “Fat2 and Lar Define a Basally Localized Planar Signaling System Controlling Collective Cell Migration” has been published in the journal Developmental Cell.
Was this helpful?