Most medical research looking to identify the mechanisms of a disease or test treatments rely on animal models. While very useful, mice for instance (a favorite lab pet for researchers) do not have nearly the same brain structure or genes as humans. Even if some genes and proteins scientists target are the same both in mice and humans, it will still be unclear whether a treatment will work for both organisms, something very important to keep in mind especially when preparing clinical trials.
The most sound alternative to this would be to culture human organs and cells for testing, and great strides forward have been made in this direction in the past decade alone. Jürgen Knoblich and Madeline Lancaster, both researchers working in labs at the Austrian Academy of Sciences, are also part of this collective scientific effort of culturing human cells for research purposes. The researchers successfully grew hundreds of 4-mm white blobs of neurons from stem cells induced from adult skin cells. Remarkably, these blogs developed specialized regions, similar to the way a human brain would develop in utero.
Previously we’ve reported how other scientists had grown human lungs, kidneys and even a heart that beats on its own (all primitive versions, not really comparable yet to the natural counterpart), while other efforts have concentrated on culturing intestines, pituitaries, and simple retinas. Testing treatments on these lab grown structures helps scientist surmount some of the challenges that come with animal models, including animal cruelty in some cases. The brain, however, is far more complex than any of these and as such is very difficult to replicate in the lab.
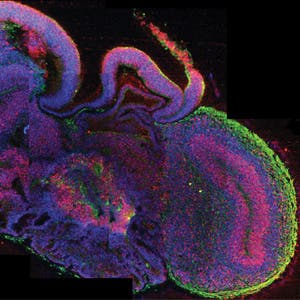
Knoblich and colleagues first collected fibroblasts (collagen generating skin cells), and induced these to become stem cells (induced pluripotent stem cells). These cells were placed in a nutrient rich gel, which allowed them to replicate into a sort of big ball of cells, akin to the embryo development stage of typical brain in utero. Some of these cells became precusor cells for neural tissue. The innovative part came in the last stage, when the researchers placed their cultured cells into a bioreactor which helped spread and distribute nutrients into the primitive brain-like tissue much in the same blood vessels would deliver the nutrients in a ‘conventional’ developing brain. The resulting brain-like structures are called cerebral organoids.
“If you’re studying brain development, you would like to see the cells develop somewhat like a brain,” says Wynshaw-Boris, who wasn’t involved in the new study. “These organoids gradually differentiate into many different types of cells. It’s not really a brain, but seems to recapitulate brain development. This is much better than any of the past efforts, in terms of the number and types of cells.”
Using the spinning nutrient distribution provided by the bioreactor, the cultured organoids reached new levels of complexity compared to previous attempts, allowing them to develop in regions similar in composition (but not in complexity) to the brain’s cerebral cortex, choroid plexus, retina, and meninges.
A brain in a petri dish
What’s most interesting about the research is that not does it only show it’s possible to culture a brain or at least a brain-like tissue in the lab, but also to use it as a model to study diseases. The researchers grew organoids using skin cells taken from a patient with microcephaly – a disorder characterized by a dramatic reduction in the size of a person’s brain. The neural tissue hared many of the trademark features of microcephaly, including reduced size. The scientists infer that in people with microcephaly, the founding neural cells don’t replicate and differentiate properly in the developing embryo, later leading to a smaller brain.
Like most current attempts at culturing human organs, the cultured organoids are simple and lack the far reaching complexity of a mature brain – they’re millions of miles apart. . A developing brain contains blood vessels that deliver nutrients and energy with which to grow, but modern science hasn’t been able to fully duplicate those blood vessels yet – the same problem that pesters other researchers hard at work culturing human hearts for instance. In fact, Knoblich believes we won’t ever be able to develop a functioning human brain, the way we’re used to thinking of it at least. Even so, it would raise some serious ethical questions. Deep down in a lab grown functioning brain’s recesses, in a cold petri dish, would a human consciousness lurk? Would you be essentially growing a human being? Such discussions have been a subject of debate for many years concerning abortions, and this new research adds further food for thought.
Findings were reported in the journal Nature.
Was this helpful?