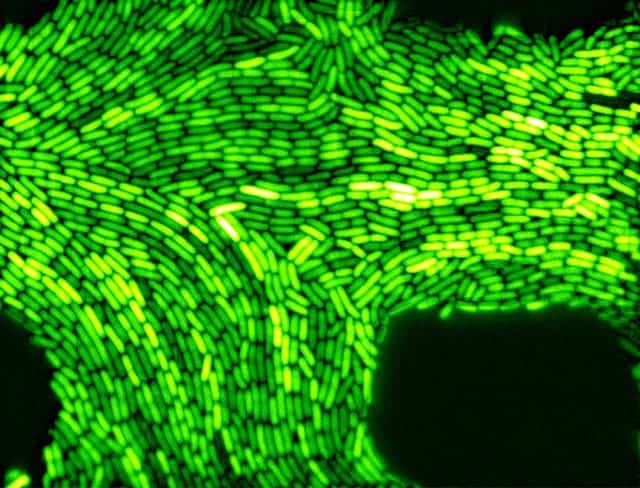
The discovery of green fluorescent proteins heralded a revolution in cell biology, enabling researchers to monitor cellular processes by applying themselves to a variety of protein and enzyme targets. Over the years, they’ve enabled thousands of successful experiments, triggering events that ultimately saved many lives.

In 1961, Osamu Shimomura and Frank Johnson, working at the Friday Harbor Laboratories of the University of Washington were working on isolating a blue bioluminescent protein from the Aequorea victoria jellyfish. When they succeeded, they called it aequorin. However, even more interesting than the aequorin, they noticed that a second protein they extracted was able to produce green fluorescence when illuminated with ultraviolet light, although it lacked the blue-emitting properties of aequorin. They called it, rather unceremoniously, Green Fluorescent Protein (GFP). To this day, we don’t know the purpose of the aequorin or the GFP in the jellyfish, but we’ve learned how to use them ourselves.

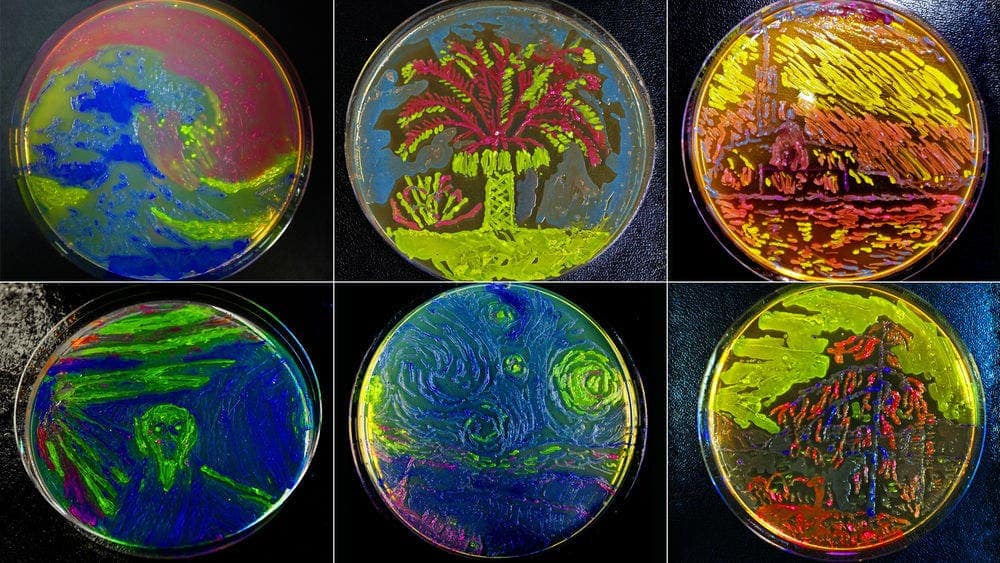
GFP can be used as a reporter gene – a gene that signals something; for example, a marker can be developed, making cancer cells (and cancer cells only) light up in green. GFP is used widely in cancer research to label and track cancer cells. But that’s not all it can do. The protein has potential uses both in regenerative medicine, and in gene therapy.
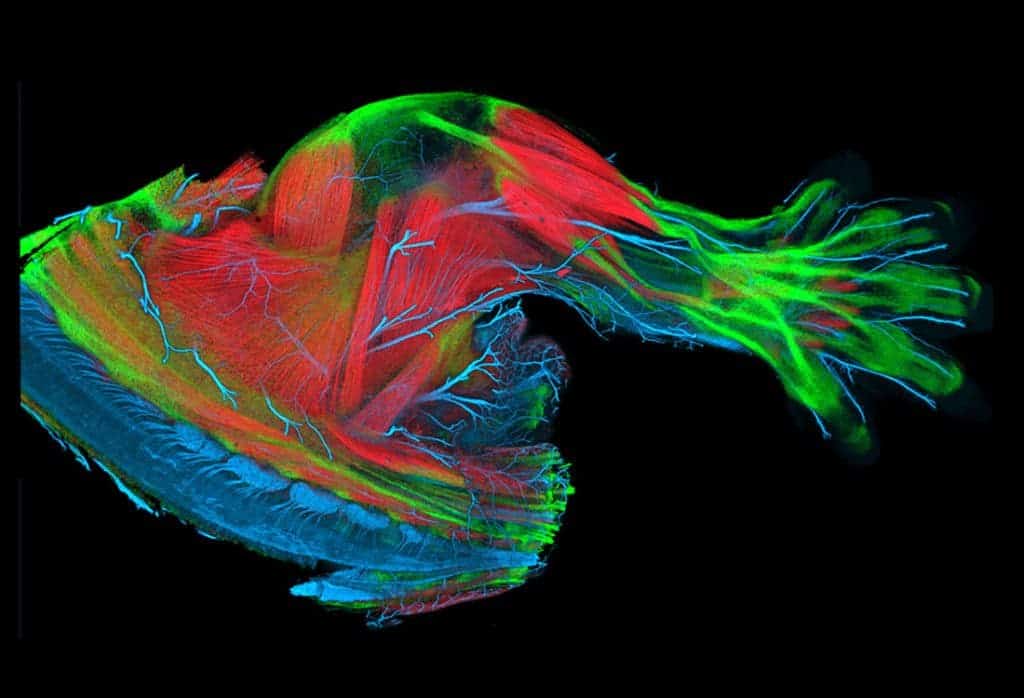
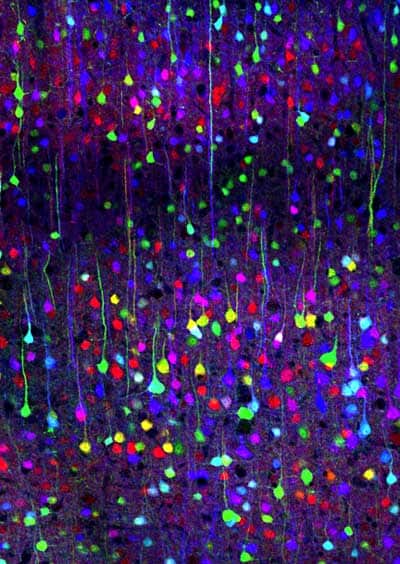
GFP can be introduced and maintained through breeding in most type of living cells – from bacteria and yeast to mammals, including humans. In fact, several transgenic pets have been developed – including a fluorescent rabbit, a group of mice and green fluorescent pigs. Creating transgenic, fluorescent fish has become so trivial that several modified species can be found on the market.


The future also has much in store for these proteins; on one hand, the goal is to expand and fine-tune the existing color possibilities derived from the jellyfish. Researchers also want to create fluorescent proteins emitting in the orange to far red regions of the visible light spectrum. Especially in mammal cells, autofluorescence and the absorption of light are greatly reduced at the red end of the spectrum – so going closer to this range would improve practical applications, including those on humans.

Secondly, the idea is to develop better biosensors based on GFP. The field of biosensors is developing at a staggering rate, and I wouldn’t be surprised to see GFP play a bigger role in the future. Martin Chalfie, Osamu Shimomura, and Roger Y. Tsien were awarded the 2008 Nobel Prize in Chemistry on 10 October 2008 for their discovery and development of the green fluorescent protein, but the future looks even brighter.
Was this helpful?